A new horizon in neurodegenerative
disease treatments
Our mission is clear:
Advancing groundbreaking science to reach a new horizon in neurodegenerative disease treatments.
We are focused on advancing innovative treatments for people living with neurodegenerative diseases, with a commitment to transparency and urgency.
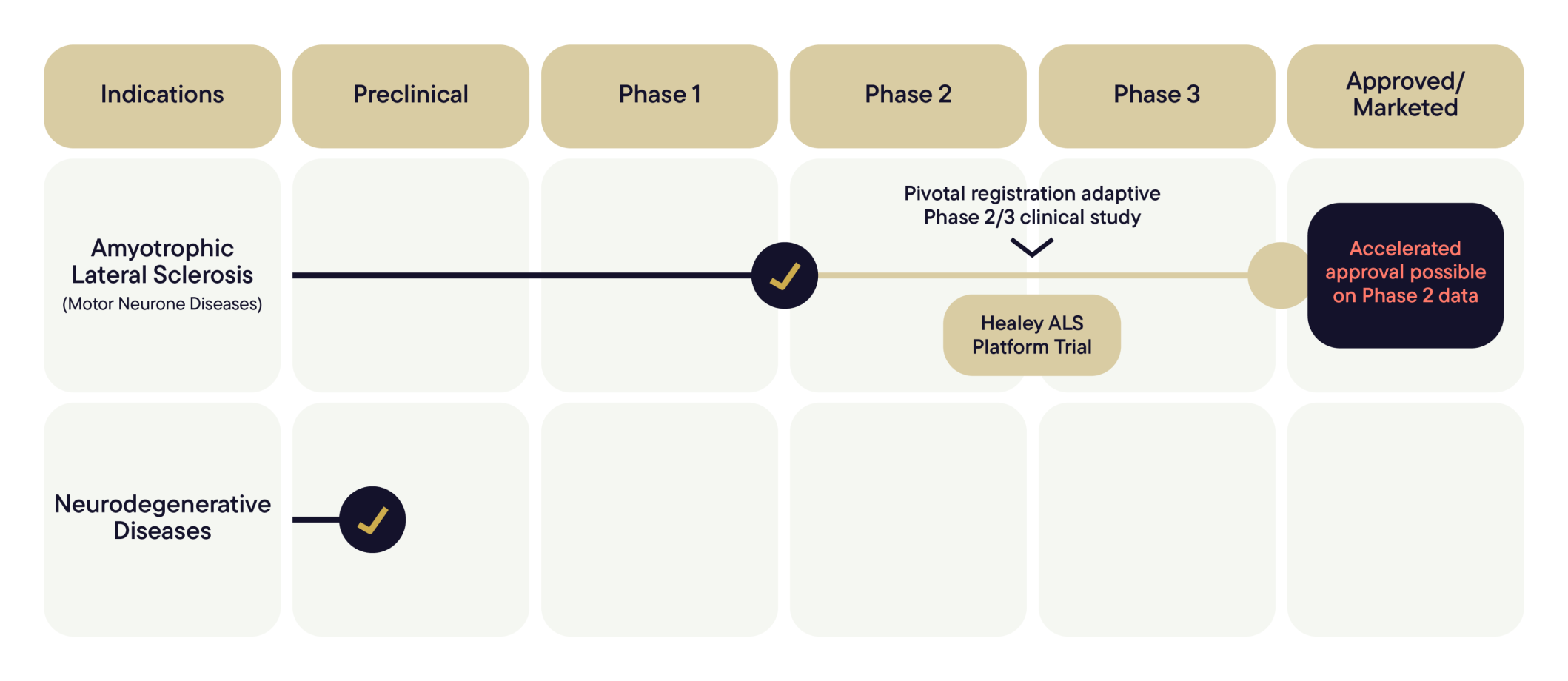
Our scientific expertise is driving this momentum. After years of research and development, we are pioneering novel applications for NUZ-001 (S-Monepantel). Our initial focus is on Amyotrophic Lateral Sclerosis (ALS), the most common form of Motor Neurone Disease (MND).
NUZ-001 (S-Monepantel) has completed Phase 1 and Open Label Extension (OLE) studies in a small cohort of people living with ALS, where treatment appeared safe and generally well tolerated, with encouraging signals observed across exploratory clinical measures.
NUZ-001 (S-Monepantel) is now being evaluated in a Phase 2/3 clinical trial as Regimen I within the HEALEY ALS Platform Trial — a major international collaboration designed to efficiently evaluate potential new treatment options for people living with ALS.
Our work also has potential relevance across other neurodegenerative diseases, supporting the broader evaluation of NUZ-001 (S-Monepantel) in these areas.

Amyotrophic Lateral Sclerosis (ALS)
ALS is a devastating neurodegenerative condition that primarily affects nerve cells that control voluntary muscles. It is characterised by severe debilitating symptoms, including muscle weakness, wasting, and paralysis. The limited treatment options available can only manage symptoms, but they cannot reverse the progression of the disease.
We’re working to bring transformative treatments to people living with ALS as quickly as possible

What’s on the horizon right now
There is no time to keep the people living with ALS waiting.
NUZ-001 (S-Monepantel) is being evaluated as a regimen “I” within the HEALEY ALS Platform Trial, an adaptive Phase 2/3 study led by the Healey & AMG Center for ALS at Mass General Brigham in collaboration with the Network for Excellence in ALS (NEALS).
The trial is conducted across multiple specialist clinical sites, with patient enrolment ongoing. This trial provides an efficient framework to evaluate potential treatments for ALS and supports ongoing regulatory engagement as the program progresses.
Product Pipeline
At Neurizon, we are dedicated to advancing innovative treatments for neurodegenerative diseases. Our approach is centred on NUZ-001 (S-Monepantel), which has shown encouraging signals in preclinical and exploratory clinical studies.

Here’s a glimpse into
our journey:
Preclinical Success
NUZ-001 (S-Monepantel) has shown significant potential in preclinical models, demonstrating its capacity to address the underlying mechanisms of neurodegenerative disease.
Phase 1 and Open Label Extension Studies
NUZ-001 (S-Monepantel) has completed a Phase 1 clinical study and subsequent 12-month open-label extension (OLE) in a small cohort of people living with ALS in Australia. Across the study period, treatment appeared safe and generally well tolerated, with no treatment-related serious adverse events reported. Top-line findings from the Phase 1 and OLE studies demonstrated encouraging trends across exploratory clinical measures, including survival, functional decline, and respiratory outcomes.
Healey ALS Platform Trial
NUZ-001 (S-Monepantel) has been accepted as Regimen I within the HEALEY ALS Platform Trial, following regulatory clearance. Patient enrolment and first dosing have now commenced, marking the initiation of the regimen within the platform trial.
Together, we’re working to make hope a reality for people living with neurodegenerative diseases
Our Team
Board of Directors
Management
Contact us
Email us: enquiries@neurizon.com
Call us: +61 (3) 9692 7222
Join us on our journey














